|
Nitrogen is in Group 5, so it has 5 outer. There is a quick way of identifying the number of valence electrons - it is the same as the Group number (not for d-block elements, though). Nitrogen has 5 electrons in its n2 (outer) shell. Learn about its discovery, history, uses, isotopes, oxidation states, and more from this web page. The number of valence electrons is the number of electrons in the outer shell, that the atom uses for bonding. It has 14 valence electrons and belongs to the p-block of the periodic table. 6d Orbital diagram of valence electrons Hence valence electron for nitrogen is 5 and for chlorine, it is 7. Most transition elements have two valence electrons, though some that have unusual electron configurations have only one. Nitrogen is a chemical element with the symbol N and atomic number 7. 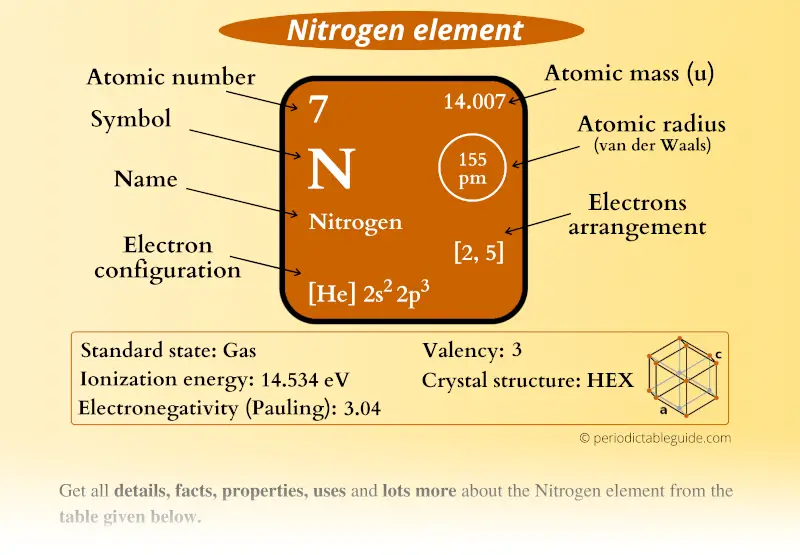 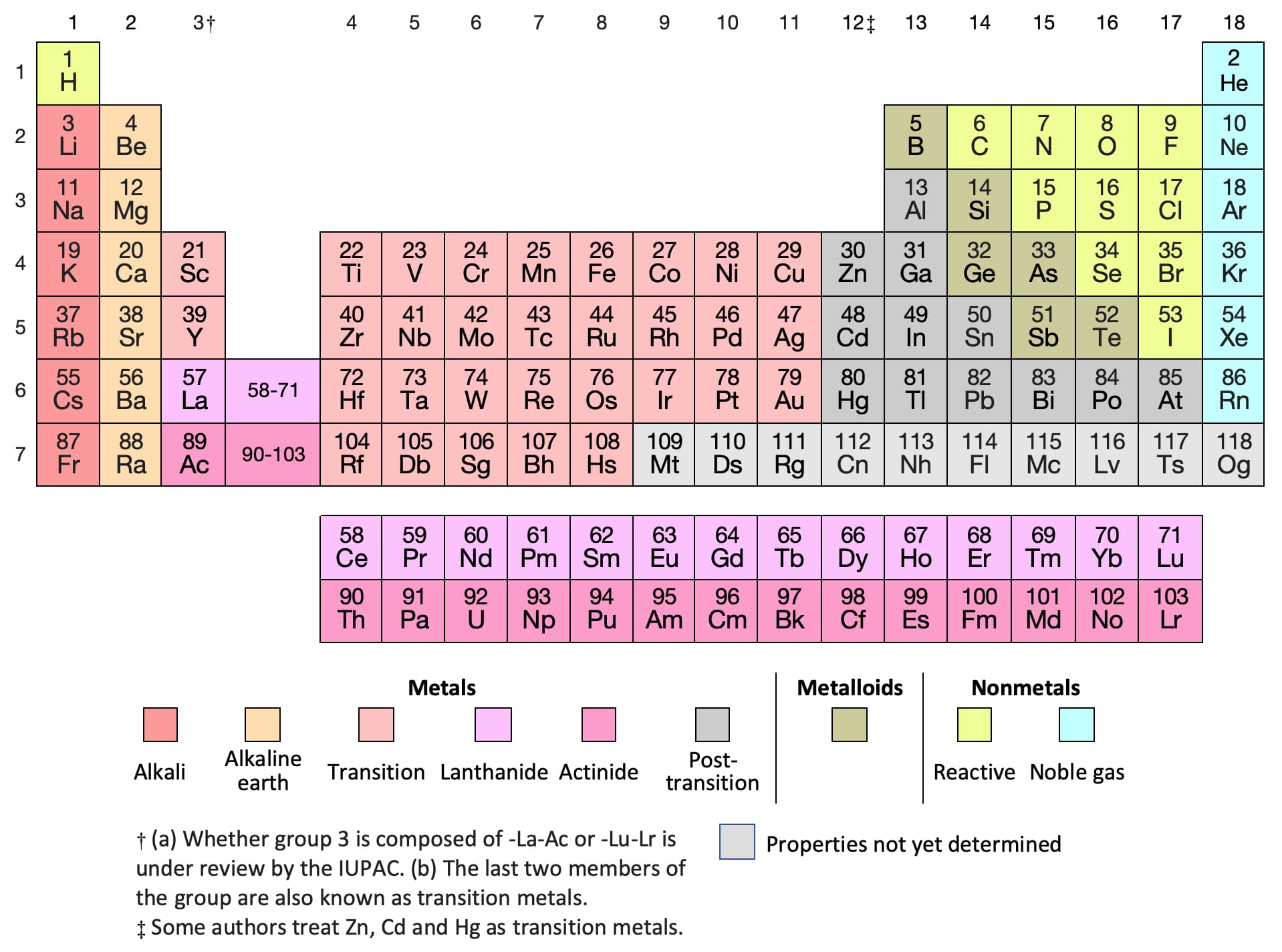 
\) (Credit: Joy Sheng Source: CK-12 Foundation License: CC BY-NC-SA 3.0(opens in new window))Įlectron dot diagrams would be the same for each element in the representative element groups.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
